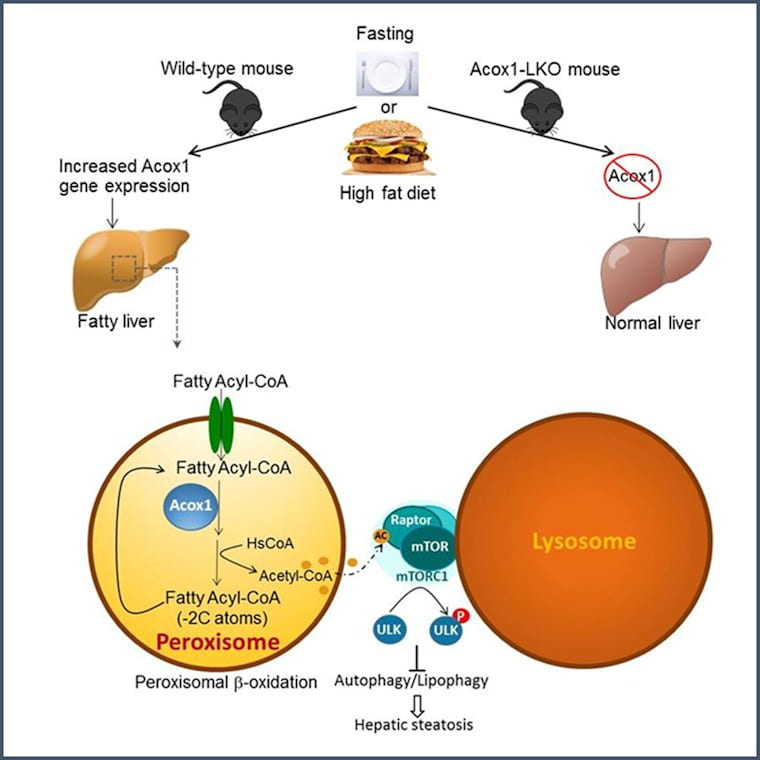
Non-alcoholic fatty liver disease (NAFLD) is the most common liver disease worldwide, with a global prevalence of approximately 25%. A new paper from the laboratory of Irfan Lodhi published in Molecular Cell on May 28 suggests that inhibition of Acox1, an enzyme that breaks down very long chain fatty acids in cellular organelles called peroxisomes, protects against fatty liver caused by starvation or high fat diet. The authors show that mice with genetic inactivation of Acox1 in the liver are protected against fatty liver due to activation of lipophagy, a subtype of self-digestion process called autophagy, which selectively degrades intracellular lipid droplets within lysosomes.
Although autophagy is activated by starvation, it is not sufficient to block the resulting fatty liver. The authors show that this is because peroxisomes signal to lysosomes to limit autophagy activation. Their research suggests that fatty acid oxidation mediated by Acox1 generates a metabolite called acetyl-CoA, which activates an autophagy inhibitory complex on lysosomes called mTORC1 (mechanistic target of rapamycin complex 1). Collectively, these studies identify a novel peroxisome-lysosome metabolic link that restricts autophagic degradation of lipid droplets. Induction of lipid autophagy through inhibition of peroxisomal acetyl-CoA production might lead to a novel treatment option for NAFLD.