Researchers led by Charles A. Harris, MD, PhD of the Division of Endocrinology, Metabolism and Lipid Research completed a research study that suggests the possibility of future cell-based therapies for patients with leptin deficiency, lipodystrophy, or other mutations in adipose-specific genes.
Collaborators included first author, Daniel Ferguson, PhD and co-authors Mitchell Blenden, Irina Hutson, Yingqiu Du and Charles A. Harris. Their research on leptin deficiency was recently featured in the Endocrine Society’s October 10th issue of Endocrine News, Mouse Study May Provide Key to Therapies for Leptin Deficiency.
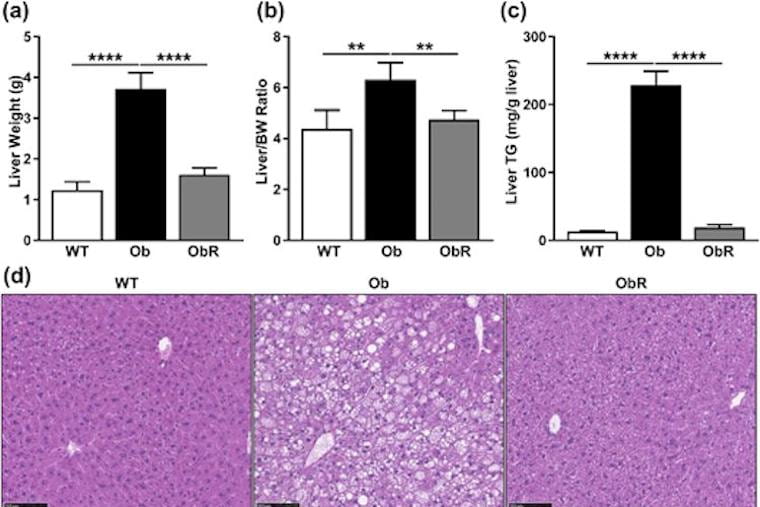
Leptin primarily produced by adipose tissue is a hormone that helps to regulate body weight. Leptin-deficient humans and mice are obese, have diabetes, are infertile, and have hepatic steatosis, according to the authors. Since leptin-replacement therapies are expensive and require daily injections, the researchers explored another approach: “Because adipocytes are the source of leptin secretion, we investigated whether mouse embryonic fibroblasts (MEFs), capable of forming adipocytes, could be injected into ob/ob mice and prevent the metabolic phenotype seen in these leptin-deficient mice,” they write.
In the study, the ob/ob mice were injected with mouse embryonic fibroblasts (MEFs) and then compared to nontreated ob/ob mice. The treated mice (obRs) had significantly lower body weight, less hepatic steatosis (fatty liver), showed greater glucose tolerance and had more energy expenditure.
Their publication based on this research: Mouse Embryonic Fibroblasts Protect ob/ob Mice From Obesity and Metabolic Complications